Most lists of the best peptides for fat loss blur two very different categories: FDA-approved obesity drugs with large clinical programs, and off-label or research compounds with narrower evidence and murkier regulation. That’s the gap that matters. If you don’t separate approved therapies from experimental options, you can’t judge risk, expected results, or what “success” should realistically look like.
The current peptide conversation also skips a practical problem. People can usually find a list of names, but they rarely get help translating those names into an executable routine: weekly versus daily dosing, adherence risks, reconstitution errors, missed-dose patterns, or when a protocol deserves a rethink. That matters just as much as the molecule itself.
This review keeps the categories clean. It starts with approved medications that have the strongest fat-loss evidence, then moves into condition-specific peptides and finally into the off-label stack that gets discussed heavily in wellness circles despite limited human fat-loss data. If you’re also comparing the major GLP-1 options directly, Mounjaro vs Ozempic for Weight Loss is a useful companion read.
Table of Contents
- 1. Zepbound (tirzepatide)
- 2. Wegovy (semaglutide)
- 3. Saxenda (liraglutide 3 mg)
- 4. Egrifta SV / Egrifta WR (tesamorelin)
- 5. IMCIVREE (setmelanotide)
- 6. Myalept (metreleptin)
- 7. CJC-1295 + Ipamorelin (growth-hormone secretagogue stack)
- Top 7 Fat-Loss Peptides Comparison
- Managing Your Protocol From Theory to Practice
1. Zepbound (tirzepatide)
Tirzepatide sits at the top of most evidence-based rankings because it’s an FDA-approved drug for chronic weight management and because the clinical results currently outpace the older GLP-1 standard. In a 2024 phase 3 trial, adults with overweight or obesity averaged 20.9% weight reduction at 36 weeks on once-weekly tirzepatide, rising to 26.4% at 52 weeks for those who continued treatment, while participants switched to placebo regained weight according to the GoodRx review of peptides for weight loss.
That result changes how to think about “best peptides for fat loss.” The key distinction involves more than appetite control versus fat burning. It’s whether a peptide has replicated, large-scale human outcomes that define an expected range of response.
Why tirzepatide currently sets the bar
Tirzepatide is a dual GIP/GLP-1 receptor agonist, which gives it a broader metabolic mechanism than semaglutide’s GLP-1-only approach. In practice, that means it’s not just another injectable peptide in the weight-loss conversation. It’s the benchmark approved option for people prioritizing maximum average efficacy from a mainstream prescription pathway.
A second reason it ranks first is regulatory clarity. Zepbound is a branded, approved medication with a labeled indication, established manufacturer information, and prescriber familiarity. That’s a very different risk profile from clinic stacks or “research” products. For readers sorting approved peptides from the rest, this overview of FDA-approved peptide drugs helps frame where tirzepatide belongs.
Practical rule: If your first question is “Which peptide has the strongest mainstream evidence for total body-weight reduction?”, approved tirzepatide is the cleanest answer on current data.
Where it fits best
Zepbound makes the most sense for people who want the strongest evidence-backed option and can tolerate the tradeoffs that come with the GLP-1 class and related agents.
- Best fit: People prioritizing large-trial efficacy and a labeled obesity indication can review the official product overview at Zepbound.
- Main limitation: Gastrointestinal side effects remain a practical barrier, and labeled contraindications matter.
- Real-world consideration: Coverage, supply, and brand-only purchasing issues can shape access even when the clinical case is strong.
2. Wegovy (semaglutide)

Semaglutide remains one of the best peptides for fat loss because it combines FDA approval, a long clinical record, and highly reproducible weight-loss outcomes in obesity care. In the STEP 1 phase 3 trial involving 1,961 adults with overweight or obesity, weekly 2.4 mg semaglutide plus lifestyle intervention produced an average 14.9% body-weight loss over 68 weeks, compared with 2.4% for placebo, according to Medical News Today’s review of peptides for weight loss.
That kind of evidence makes Wegovy the “reference drug” for many comparisons, even when tirzepatide posts stronger averages. Semaglutide has become the standard many clinicians, patients, and analysts use to judge whether a newer peptide is meaningfully better or just newer.
What the evidence says
The most useful semaglutide detail isn’t just the average loss. It’s response distribution. In the same trial, large portions of participants crossed meaningful weight-loss thresholds, which tells you semaglutide isn’t relying on a few extreme responders to lift the average. That matters when evaluating predictability rather than hype.
Semaglutide also benefits from clear regulatory status. It’s FDA-approved for chronic weight management in adults meeting BMI criteria and was later extended to adolescents meeting the label criteria, which strengthens its standing as a medically established option rather than a wellness-market trend.
If someone is using compounded or reconstituted semaglutide in another context, execution quality becomes a separate issue from molecule quality. This guide on how to reconstitute semaglutide is relevant because a dosing mistake can distort both tolerability and perceived effectiveness.
Semaglutide’s biggest advantage isn’t novelty. It’s that clinicians already know how to use it, titrate it, and monitor its common failure points.
Why some patients still choose semaglutide first
Tirzepatide may lead on average outcomes, but Wegovy still wins many real-world decisions because the evidence base is broad, the prescribing comfort level is high, and the weekly schedule is simple.
- Strength: It’s an established weekly GLP-1 option with strong obesity-trial support and official product information at Wegovy.
- Tradeoff: GI side effects and class warnings still shape eligibility.
- Analyst view: If tirzepatide is the current efficacy leader, semaglutide is still the most proven “default comparator” in peptide-based obesity treatment.
3. Saxenda (liraglutide 3 mg)
Saxenda rarely tops today’s lists, but leaving it out would distort the market. It’s an FDA-approved GLP-1 receptor agonist for chronic weight management, and its continued relevance comes from familiarity, access pathways, and the fact that some patients still do better with a daily titration model than with a weekly injection.
Many “best peptides for fat loss” articles become too simplistic; they assume the highest-efficacy drug is automatically the best choice for every patient. That isn’t how obesity treatment works in practice. Tolerance, insurance behavior, prescriber habits, and patient preference still matter.
Why Saxenda still matters
Liraglutide 3 mg has been in clinical use long enough that many clinicians are comfortable with its titration pattern, side-effect counseling, and monitoring. That doesn’t make it the strongest option on average. It makes it a workable alternative when a weekly GLP-1 isn’t available, tolerated, or preferred.
A daily injection can also create a different behavioral dynamic. Some patients dislike it. Others prefer the routine because it gives them a stronger sense of control and makes it easier to connect symptoms with dose timing.
A lower-ranked peptide can still be the right peptide if access and adherence are better.
Tradeoff to understand
Saxenda’s core weakness is straightforward. It generally delivers less average weight loss than the newer weekly agents discussed above. That doesn’t need exaggeration. This means Saxenda now occupies more of a fallback or fit-dependent role rather than the lead slot.
- Best fit: People who need an FDA-approved alternative and can work with a daily pen can review the official product page for Saxenda.
- Advantage: Long market history and a predictable titration routine.
- Constraint: Daily injections create more opportunities for adherence drift, especially over long treatment periods.

4. Egrifta SV / Egrifta WR (tesamorelin)
Tesamorelin belongs on this list for a different reason than tirzepatide or semaglutide. It isn’t a general obesity drug. It’s an FDA-approved growth hormone-releasing hormone analog indicated to reduce excess visceral abdominal fat in adults with HIV-associated lipodystrophy.
That narrower indication is exactly why it deserves a place. It shows that “fat loss” is not one clinical target. Reducing visceral adipose tissue in a specific disease context is different from pursuing broad chronic weight management in the general population.
A targeted option for visceral fat in a specific condition
Tesamorelin stands out because its approved use is tightly connected to body-fat distribution rather than standard obesity labeling. For the right patient population, that specificity matters more than mass-market popularity.
This is also where many readers miss a key distinction. A peptide can be highly relevant to fat reduction without being appropriate for general-purpose slimming or fitness use. Tesamorelin is a good example of a peptide whose value rises when the patient profile becomes more precise.
- Clinical role: It’s approved for a narrow condition involving excess visceral abdominal fat, with details available through Egrifta SV.
- Why it ranks here: The mechanism and indication are specific, legitimate, and clinically meaningful.
- Limit: That specificity also sharply limits who should consider it.
Why it shouldn’t be treated like a general fat-loss peptide
Tesamorelin gets miscast online as a belly-fat shortcut. That framing strips out the reason it was approved in the first place. Outside the labeled HIV-associated lipodystrophy context, readers should be cautious about transferring disease-specific logic into general obesity claims.
5. IMCIVREE (setmelanotide)
Setmelanotide is one of the clearest examples of why “best” depends on biology. For common obesity, it isn’t a general first-line answer. For certain rare, genetically confirmed forms of obesity, it can be highly relevant because it targets the melanocortin-4 receptor pathway directly.
That makes IMCIVREE one of the most mechanism-specific entries on this list. It isn’t broad. It is precise.
Mechanism first, not mass-market use
Setmelanotide’s place in fat-loss treatment depends on genotype, not on wellness trends or social-media popularity. If someone doesn’t have one of the rare, genetically confirmed conditions covered by the label, IMCIVREE is not built for their use case.
This is an important analytical dividing line. Some peptides are “best” because they help the largest number of people. Others are “best” because they precisely match a narrow biological problem.
The right way to rank setmelanotide is not by mainstream demand. It’s by fit to the labeled genetic disorder.
How to think about its place on this list
IMCIVREE shouldn’t be compared casually with Wegovy or Zepbound as if all three are interchangeable obesity drugs. They’re operating in different clinical lanes.
- Ideal use case: Patients with qualifying, genetically confirmed obesity conditions can start with the manufacturer information at IMCIVREE.
- Strength: Disease-mechanism specificity.
- Limitation: Genetic testing, specialty access, and authorization requirements sharply narrow eligibility.
6. Myalept (metreleptin)
Myalept is another peptide that doesn’t belong in a casual “fat burner” bucket. It’s a leptin analog approved to treat complications of generalized lipodystrophy, not common obesity. That distinction matters because this therapy addresses leptin deficiency in a rare disease state rather than offering a conventional weight-loss strategy for the broader population.
So why include it in a list of the best peptides for fat loss? Because readers who want a serious evidence review should see the full map, not just the commercial center of the market. Myalept is part of that map.
A replacement therapy, not a mainstream weight-loss drug
Metreleptin is best understood as a condition-specific replacement therapy with metabolic goals. It sits closer to endocrine correction than to the mainstream anti-obesity model represented by GLP-1 and GIP/GLP-1 drugs.
That changes the evaluation standard. You wouldn’t rank Myalept based on whether it competes with Wegovy for average body-weight reduction in typical obesity. You’d rank it based on whether it meaningfully addresses the complications of generalized lipodystrophy under structured specialist oversight.

Why inclusion still matters
Myalept is REMS-restricted, specialty-distributed, and tightly supervised. Those access barriers aren’t incidental. They reflect the seriousness of the condition and the need for controlled use.
- Appropriate context: It’s the FDA-approved pharmacologic option for generalized lipodystrophy, with professional information at Myalept.
- What that means for most readers: It’s relevant among the available peptides, but not as a general obesity solution.
- Analyst takeaway: Approval status alone isn’t enough. You also need to ask, “Approved for whom?”
7. CJC-1295 + Ipamorelin (growth-hormone secretagogue stack)

This stack is where the article shifts from approved medicine to the off-label and research side of the peptide world. CJC-1295 plus ipamorelin is widely discussed in wellness, fitness, and biohacking circles because the pairing is thought to support pulsatile growth hormone signaling through combined GHRH and GHRP action. But the evidence position is much weaker than the approved drugs above.
The most important fact isn’t a dramatic outcome claim. It’s the gap in the evidence base. Available peptide content often describes this stack as synergistic, yet the practical guidance that users need is largely missing. The Stem Cell Miami overview of weight-loss peptides reflects that gap by emphasizing what peptides may do without solving real scheduling questions such as timing relative to meals or training, cycling structure, injection rotation, or how to run a multi-peptide stack consistently.
Why this stack remains popular despite thin evidence
Popularity here comes from mechanism and anecdote, not from the kind of large, obesity-focused human trial record that supports tirzepatide or semaglutide. Another gap is individual response variability. Commentary around peptide stacks often ignores that adherence failures, missed doses, incorrect reconstitution, storage issues, and weak self-monitoring may be the main reasons a protocol underperforms, a problem highlighted in Lainey Younkin’s discussion of fat-burning peptides.
That makes this stack harder to evaluate accurately. If someone reports “it worked,” was it the peptide, better sleep, more training discipline, reduced calories, or simple consistency after weeks of chaos? In many cases, no one knows.
For readers comparing versions, CJC-1295 DAC vs no DAC is one of the practical distinctions that changes how a protocol is organized.
Field note: The execution burden is much higher with off-label stacks because the user often has to solve dosing, scheduling, and tracking problems without the support structure that comes with an approved branded drug.
The real execution problem
The stack’s biggest weakness isn’t just regulation. It’s operational fragility.
- What supports interest: A plausible GH/IGF-1 rationale and practitioner use in some clinic settings.
- What limits confidence: Limited controlled human fat-loss outcomes for the general population.
- Regulatory concern: U.S. compounding and supply issues add uncertainty, and the FDA context is reflected in this FDA presentation document.
Top 7 Fat-Loss Peptides Comparison
| Intervention | Complexity / Process 🔄 | Resources & Access ⚡ | Expected outcomes ⭐📊 | Ideal use cases 💡 | Key advantages ⭐ |
|---|---|---|---|---|---|
| Zepbound (tirzepatide) | Weekly injection; standard clinic monitoring; label contraindications (e.g., endocrine tumors/MEN2) | Prescription-only; coverage and cash pricing vary; multi‑dose KwikPen improves convenience; watch for counterfeits | Among highest average weight loss in phase 3 trials; metabolic benefits reported | Adults with obesity/overweight plus comorbidity seeking maximal weight loss | High trial efficacy; clear labeled indication; strong prescriber familiarity |
| Wegovy (semaglutide) | Weekly injection or oral tablet option; routine monitoring; class boxed warnings apply | Broad availability; manufacturer programs and evolving pricing; oral formulation expands options | Strong long‑term weight loss and cardiometabolic benefits across programs | Patients preferring an established GLP‑1 with flexible formulation choices | Extensive clinical track record; multiple doses/formulations |
| Saxenda (liraglutide 3 mg) | Daily injection with stepwise titration; predictable clinic workflow | Prescription with patient‑support programs; daily dosing affects adherence and cost | Moderate weight loss on average versus weekly agents | When weekly GLP‑1s are not tolerated, not covered, or patient prefers daily titration | Predictable titration schedule; long market history and clinician familiarity |
| Egrifta SV / WR (tesamorelin) | Specialty indication workflow; formulation‑specific reconstitution/dosing; HCP support portals | Specialty distribution for HIV lipodystrophy; potentially high cost and variable insurer support | Demonstrated reductions in visceral adipose tissue (VAT) and metabolic improvements in labeled population | Adults with HIV‑associated lipodystrophy and excess visceral fat | Only FDA‑approved therapy targeting VAT in HIV lipodystrophy; protocolized data |
| IMCIVREE (setmelanotide) | Requires genetic testing and specialty authorization; specialty distribution | Restricted to genetically eligible patients; high specialty‑drug cost and payer navigation | Substantial weight and hunger reductions for specific MC4R‑pathway deficiencies | Patients with confirmed POMC, PCSK1, LEPR or other labeled genetic obesity causes | Disease‑mechanism‑specific therapy with meaningful responses in eligible patients |
| Myalept (metreleptin) | REMS‑restricted; structured safety monitoring and specialty oversight | Specialty pharmacy access for generalized lipodystrophy; high cost and frequent prior authorizations | Improves glycemia/triglycerides and reduces ectopic fat in generalized lipodystrophy | Patients with congenital or acquired generalized lipodystrophy and leptin deficiency | Only approved pharmacologic option for GL with formal safety program |
| CJC‑1295 + Ipamorelin (GHS stack) | Off‑label; variable compounding/quality; regulatory and monitoring uncertainty | Not FDA‑approved for fat loss; supply and quality inconsistent; potential legal/regulatory issues | Limited, heterogeneous evidence for fat‑loss; anecdotal reports of improved recovery/body composition | Experimental use in supervised wellness or research settings; not standard obesity therapy | Reported flexible dosing and some practitioner‑observed body‑composition effects under supervision |
Managing Your Protocol From Theory to Practice
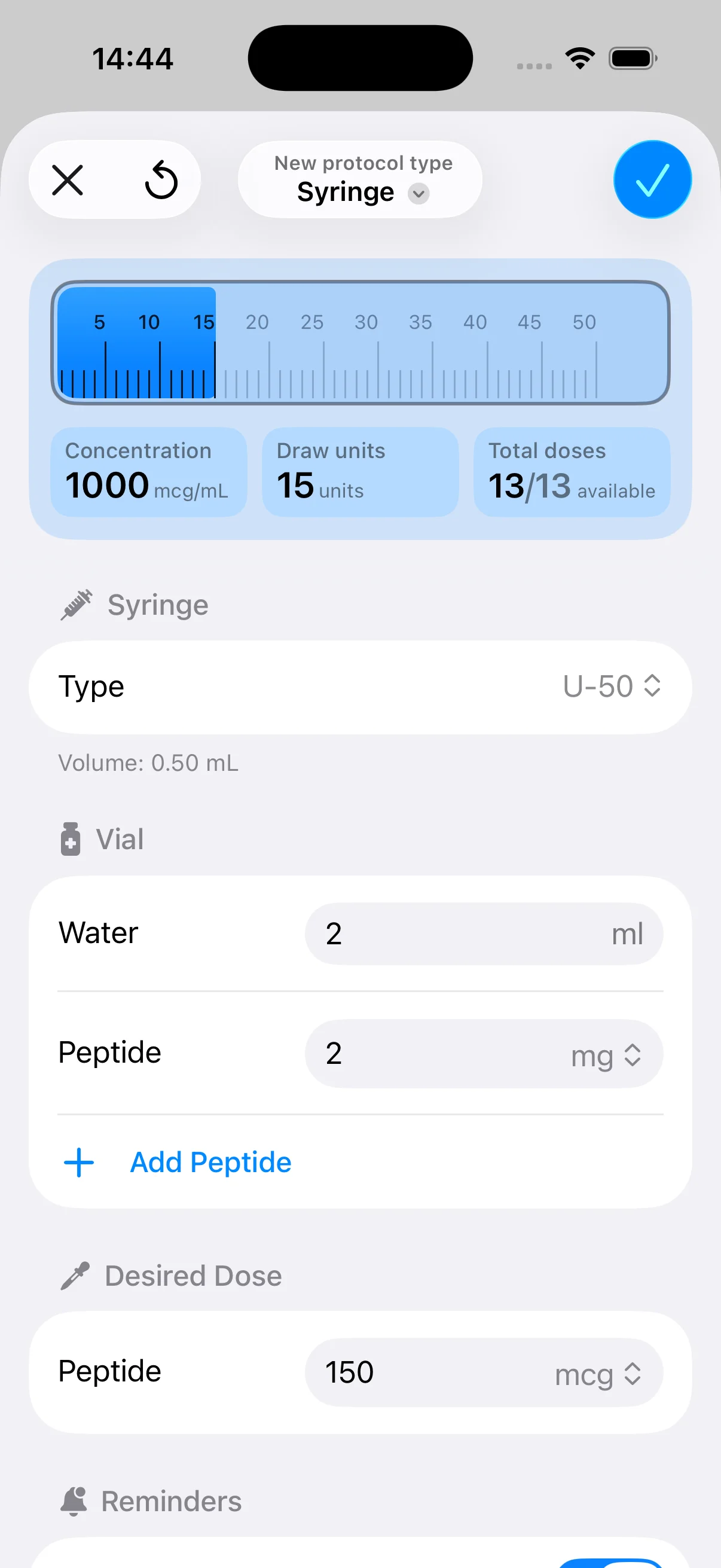
Knowing which compounds rank highest is only part of the job. The harder part is running a protocol correctly for long enough to judge it fairly. That’s true for weekly approved medications, daily injectables, and even more so for off-label stacks that require reconstitution, cycle planning, and more detailed self-monitoring.
The evidence reviewed above points to a practical conclusion. Approved options such as tirzepatide and semaglutide have stronger clinical footing, but even those drugs depend on adherence. The off-label side adds another layer of difficulty because mainstream content rarely answers execution questions clearly. The biggest gaps aren’t always about mechanism. They’re about timing, consistency, dose logging, and recognizing when a response pattern is poor enough to revisit the plan.
That’s where tools become relevant. A scheduling and dosing workflow can’t make an ineffective protocol effective, but it can reduce preventable errors. For someone following a daily, weekly, or cycled regimen, that means fewer missed doses, fewer handwritten calculations, and a cleaner record of what happened. If you’re also dialing in nutrition alongside a peptide plan, a Macro Calculator for Weight Loss can complement the medication side of the equation.
PepFlow is one example of that operational layer. Based on its published product description, it helps users calculate doses, configure vial concentration details, build cyclical schedules, and log administration history. That doesn’t replace clinical judgment, but it does address a real gap that keeps surfacing across peptide use: turning theory into repeatable execution.
For discerning readers, the main takeaway is simple. The best peptides for fat loss aren’t all “best” in the same way. Tirzepatide and semaglutide lead for broad, approved obesity treatment. Tesamorelin, setmelanotide, and metreleptin matter because they serve narrower disease-specific roles. CJC-1295 plus ipamorelin remains interesting, but the evidence and regulatory footing are much less secure. Once you separate those categories, the decision framework becomes clearer. Start with evidence strength, then regulatory status, then fit for your condition, then your ability to execute the regimen consistently.
If you’re managing peptide schedules, dose calculations, or cyclical protocols and want a simpler way to stay organized, PepFlow is built for that planning layer. It helps with dosage math, protocol scheduling, reminders, and adherence tracking, which can make structured routines easier to run consistently.